It is hard to imagine a more fundamental as well as complicated biological object than a living cell. Currently, optical microscopy techniques are the primary method for cell surface visualization, with microscopic characteristics of cells traditionally used for diagnosis and classification of cancers. AFM is also applied in this field as a separate technique or in combination with optical microscopy. In this field Atomic Force Microscopy features not only high-resolution imaging of cellular structures below the optical limit (that being quite "natural" for this method), but also evaluation of micromechanical properties of the cell and ability to monitor cell dynamics and processes running in it even in real time. At present, no other microscopic techniques are able to provide directly both structural information of a biological sample and related functional information at such high spatial resolution.
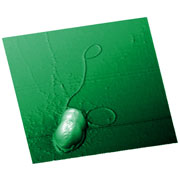
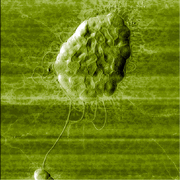
| Fig. 1. Tapping mode AFM image of a bacteria cell (a) and aggregate of cells (b) obtained wtih a HQ:NSC18 AFM probe. Image courtesy of Prof. I. V. Yaminsky, MSU (Moscow). |
 |
 |
| (a) AFM snap image of cell division (Escherichia coli bacteria cell). Scan size 6.5x6.5µm. |
(b) AFM height image of a monolayer aggregate of Escherichia coli bacteria cell (55 cells). Mica substrate. Scan size 13x13µm. |
Direct imaging of fixed or living cells and sub-cellular structures provides important information about the architecture of the membrane, organelles and cytoskeleton of cells. AFM offers a unique opportunity to image, localize and identify integral membrane proteins at the surface of living cells. Although one couldn't avoid staining or fluorescent labeling to mark the proteins of interest due to indiscriminative nature of AFM probing relative to chemical composition and nature of the objects, further improvements and extensions promise this problem to be solved. The other important function of AFM is its ability to examine mechanical properties of tiny objects. Information about the micromechanical properties is quite important for cellular systems because it helps to understand cell architecture and it's functions. Local elastic properties of a cell can be quantitatively derived from the force versus distance (F-S) curves obtained at fixed surface points using AFM. Spatial variations of mechanical properties are also reflected in the image contrast helping to visualize different cell components.
The capabilities of AFM in studies of cells are extremely broad, yet their practical realization is far from being routine and easy-to-use method. Using AFM cells can be imaged directly but sample preparation might require a fixation of the cells to a substrate. AFM study of cells in most native physiological media such as aqueous solutions is also a more difficult procedure than in air. The imaging of cells due to their large dimensions and soft nature might takes some time because scanning rates in studies of cells with the contact and oscillatory modes are quite low (0.2-0.5Hz). The larger sizes of certain cells could also necessitate the use of AFM scanners with large Z-range and also AFM probes in which the AFM tip is longer than the one of AFM probes applied for studies of flat objects. All these questions are under consideration by AFM manufacturers and, hopefully, researchers will get most appropriate microscopes in the near future.
Another major AFM application in cell studies is real-time monitoring of living cells dynamics, intercellular interactions and response to internal and external perturbations. The main problem in monitoring dynamic behavior of the cell is diminishing of perturbations induced by an AFM probe during scanning process as well as maintaining stable environmental conditions for both temperature and pH value. Another technical challenge is to make higher temporal resolution since time to acquire a full scan of a living cell often exceeds the characteristic alterations happening in it.

