Molecular self-assembly is the assembly of molecules without guidance or management from an outside source. There are two types of self-assembly, intramolecular self-assembly and intermolecular self-assembly. Most often the term molecular self-assembly refers to intermolecular self-assembly, while the intramolecular analog is more commonly called folding.
Molecular self-assembly is directed through noncovalent interactions, such as hydrogen bonding, metal coordination, hydrophobic forces, van der Waals forces, pi-pi interactions, and/or electrostatic effects. Simple examples include the formation of a micelle or a Langmuir monolayer by surfactant molecules in solution. More advanced examples of supramolecular assemblies demonstrate that a variety of different shapes and sizes can be obtained using molecular self-assembly.
In the speculative vision of molecular nanotechnology, microchips of the future might be made by molecular self-assembly. Using molecular self-assembly the final (desired) structure is programmed in the shape and functional groups of the molecules. Self-assembly is referred to as a 'bottom-up' manufacturing technique in contrast to a 'top-down' technique such as lithography where the desired final structure is etched from a bulk material
Fig. 1 presents AFM images taken by a Nanoscope IIIa MultiMode AFM and a Hi'Res-C AFM probe. The images are of self-assemblies of palindromic rotaxane molecules formed on HOPG (a, b). Ordered structures were found in ultrathin films (c), and with an increase in film thickness the top layer becomes grainy. Hi'Res-C images of the grain structure show very high resolution. Images (d) and (e) compare routine height scans of the thick film obtained by Hi'Res-C and standard silicon AFM tips.
| Fig.1. Images courtesy of Dr. S. Magonov (Veeco Instruments). Samples courtesy of Stoddart Supramolecular Group, Department of Chemistry and Biochemistry, University of California, Los Angeles. |
 |
 |
 |
| (a) Height image of the ultrathin film, Hi'Res-C AFM probe. Scan size 500nm, height 5nm. |
(b) Phase contrast of the same area, Hi'Res-C AFM probe. Z-scale is 30°. |
(c) Height image of the thin film at high magnification (scan size 70nm), Hi'Res-C AFM probe. |
 |
 |
|
| (d) Height image of the thick film, Hi'Res-C AFM probe. Scan size 250nm. Height 3nm. |
(e) Height image of the same thick film, standard AFM probe width R<10nm. Scan size 250nm. Height 3nm. |
|
Generally, organic monolayers on hard substrates have enough mechanical stiffness to be examined with the standard silicon AFM probes with stiffness of ~40N/m. The use of softer AFM probes might be necessary for samples with increasing layer thickness if topmost topography is of primary interest. Thicker layers might be necessary to examine with stiff probes when one can visualize heterogeneities and to reveal sub-surface morphology that requires a tip penetration into material.
A Langmuir-Blodgett film is a monolayer of an organic material, transferred from the surface of a liquid onto a solid by immersing (or pulling) the solid substrate into (or from) the liquid. An additional monolayer can be added with each immersion or emersion step, thus films with very accurate thickness can be formed. The monolayers are usually composed of polar molecules with a hydrophilic head and a hydrophobic tail (example: fatty acids).
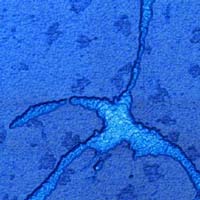
Magnetic iron-containing nanoparticles in Fig.2 are synthesized in a mixed Langmuir monolayer (stearic acid + Fe(CO)5) at the gas/liquid interface via ultraviolet decomposition of iron pentacarbonyl.
| Fig.2.Image courtesy of Radmir Gaynutdinov (A.V. Shubnikov's SPM group, Crystallography Institute, RAS). Sample courtesy of G. B. Khomutov's group, Physical Faculty, MSU. |
 |





